Who we are
The joint Data Controllers for TYPPEX are Cambridgeshire and Peterborough NHS Foundation Trust and the University of Cambridge. This means that both organisations are responsible for looking after your information and using it properly.
As a data processor, Norwich Clinical Trials Unit (at the University of East Anglia) will be collecting and storing study data on behalf of the sponsors. This means we follow the instructions of these organisations when handling certain personal data.
The TYPPEX study team abides by the University’s data protection-related policies, listed on our webpages.
Further details, including information about your rights
You can find general information about your rights, and the University’s data protection practices and obligations, in this web page.
How to contact us
You can contact the study office on 07547935702 or typpex@medschl.cam.ac.uk.
You can contact the University’s Data Protection Officer at data.protection@admin.cam.ac.uk or by telephone on +44 (0) 1223 764142.
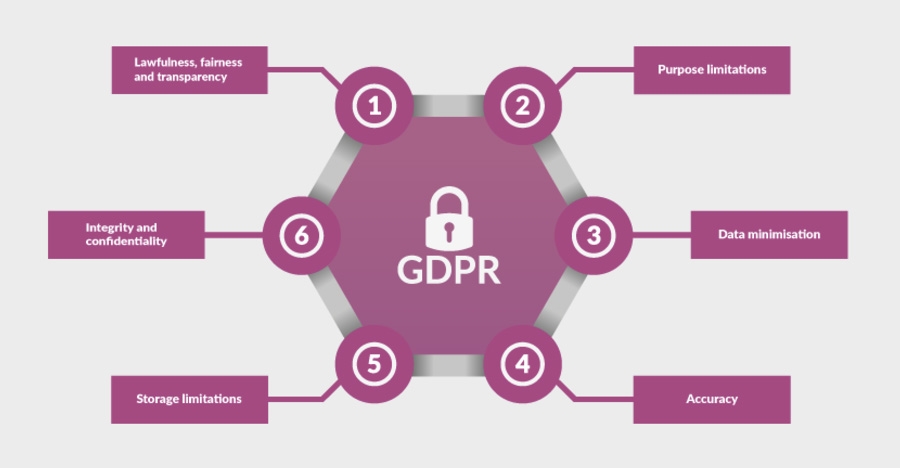
What we do with personal data
The TYPPEX study team hold personal information about 3 different groups of people:
- Research management and administrative contacts at organisations that we interact with to perform our research
- Staff at participating NHS Talking Therapies services who participate in the study’s intervention and control activities
- Service users at participating NHS Talking Therapies services
What information is held, why, and how it will be processed, differs for each of these groups.
For research management and administrative contacts
The TYPPEX study team needs to know your name, place of work, and contact details (a phone number and email address) to enable the team to administer the study from start to completion.
For NHS Talking Therapies staff
The TYPPEX study team only holds your personal data if you have agreed to take part in the study as a participating therapist. Information about the data we hold and how it is used is available in your Participant Information Sheet.
For consented service user participants
The TYPPEX study team only holds your identifiable personal data if you have agreed to take part in the study. Information about the data we hold and how it is used is available in your Participant Information Sheet.
All information which is collected about you during the course of the research will be kept strictly confidential, stored in a secure and locked office, and on a password protected database. We will ask you to consent to the research team being able to access your NHS Talking Therapies service case notes and link this data to your questionnaire responses. The data being analysed will not contain your name, NHS number or contact details.
Cambridgeshire and Peterborough NHS Foundation Trust and the University of Cambridge are joint sponsors for this study based in England. The sponsors will be using information from you and your NHS Talking Therapies service records in order to undertake this study and will act as joint data controllers. This means that both organisations are responsible for looking after your information and using it properly.
Norwich Clinical Trials Unit will be collecting and storing study data on behalf of the sponsors. The University of Cambridge, Cambridgeshire and Peterborough NHS Foundation Trust, and Norwich Clinical Trials Unit will keep identifiable information about you for 3 months after the study has finished. All non-identifiable research data will be kept securely for 10 years. After this time the data will be disposed of securely.
Your rights to access, change or move your information are limited, as we need to manage your information in specific ways in order for the research to be reliable and accurate. If you withdraw from the study, we will keep the information about you that we have already obtained. To safeguard your rights, we will use the minimum amount of personally identifiable information possible.
You can find out more about how we use your information using the following link:
https://www.medschl.cam.ac.uk/research/privacy-notice-how-we-use-your-research-data/
Use of pseudonymous NHS Talking Therapies data
During the trial we are collecting pseudonymous data about people receiving talking therapy from our participating NHS Talking Therapies services. This is to give us a complete picture of the impact of the TYPPEX intervention on therapists’ overall caseloads.
These data do not contain any information that would identify you personally, and will only be used for research purposes. The data cannot be re-identified by the research team.
If you do not want your data to be used in this way, please speak to your therapist or admin team who will document this according to local policies. You can also contact the research team directly using the details at the top of this page.